Atomic Calculator (proton-electron pair)

Fig 1. Particles
This web page and the maths associated with this calculator are copyright protected by
Keith Dixon-Roche {© 11/06/2019}
This atomic model represents the culmination of the work carried out by Keith Dixon-Roche on Isaac Newton's laws of orbital motion. It requires no obscure theories such as Relativity or Quantum Theory and needs no unification theory, but It can be mathematically verified.
The [only] two fundamental components in the universe are the proton and the electron; there are no other particles (Fig 1; see neutron below).
The atom is a collection of proton electron pairs that were fused together in galactic force-centres
Atomic Particles

Fig 2. H⁺
The proton constitutes 99.3% of all natural hydrogen gas (H⁺). It does not move unless forced to do so by impact or magnetism. Lone protons cannot absorb or transmit electro-magnetic energy. Two adjacent protons can never be adjoined (in viscous form) because their like (positive) electrical charges will always repel (Fig 2).
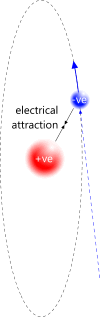
Fig 3. Proton-
Electron Pair
The electron is a much smaller particle (than the proton). It possesses perpetual kinetic energy absorbed and converted from surrounding electro-magnetic energy. Its velocity varies with the amount of electro-magnetic energy absorbed. A lone electron will travel in a straight line unless impacted by another electron or diverted by magnetism. A moving electron possesses the same angular velocity it had when ejected from its latest proton-electron partnership. In reality, electrons never actually impact, their like (negative) electrical charges will always repel.
When a proton detects a passing electron, their dissimilar charges will attract. However, the perpetual motion in an electron will never allow them to unite. The electron will simply orbit the proton. Because the electron provides its own kinetic energy, its orbital path will be circular. They will then have become a proton-electron pair (Fig 3).
As more heat (energy) is absorbed by the orbiting electron, its velocity will increase, causing its orbital radius to reduce (Fig 4), exactly as Newton described in his Principia. If sufficient energy is absorbed by the proton-electron pair, the proton and its orbiting electron will unite to be come a neutron, trapping its contemporaneous energy within. The neutron's electrical charges will be identical and opposite, cancelling out each other because electrical energy is shared, but their magnetic charges will be added together because magnetic energy accrues. The orbital radius at which this occurs is the neutronic radius and the velocity of the electron will be that of electro-magnetic radiation (c). This is the true meaning of E=mc²
The neutron is a lone particle that comprises a proton-electron pair that combined with high temperature. It possesses no electrical charge but the same magnetic charge as a proton plus an electron. It also possesses the energy generated in the proton-electron pair at the time they united.
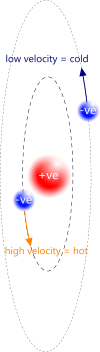
Fig 4. Orbits
The most basic atoms come in various forms, all of which are hydrogen (Fig 5):
Hydrogen (H⁺): a lone proton
Hydrogen (H): a proton-electron pair
Deuterium (D): a proton-electron pair that has trapped a neutron
Tritium (T): a proton-electron pair that has trapped two neutrons
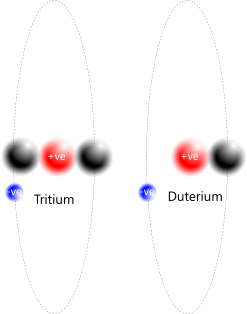
Fig 5. Hydrogen Atoms
An atom with an atomic number greater than 1 can only be created from Deuterium and Tritium. Natural hydrogen (H & H⁺) cannot be combined into larger atoms because of the coupling ratio.
The proton-neutron collection of any atom is called its nucleus.
Fusion
To create a larger atom, you simply push the nucleus of one atom inside the innermost electron shell of another atom. This can only occur if sufficient force is generated to overcome the repulsion from competing electron charges and the process is called fusion.
Electro-Magnetic Energy
A proton-electron pair is a negative charge orbiting a positive charge both of which have magnetic charges, just like a motor or generator. In this way, the proton-electron pair can generate and emit electro-magnetic energy (Fig 6). This is what we see and feel as light and heat and is the means whereby energy is transferred (between electrons).
It is important to understand that stars are not created from hydrogen and that all fusion, which requires the input of gravitational energy, only occurs in the universe's largest celestial bodies; the lighter elements are created in galactic force-centres and the heaviest elements are created in the ultimate-body.
The Atomic Calculator - Technical Help
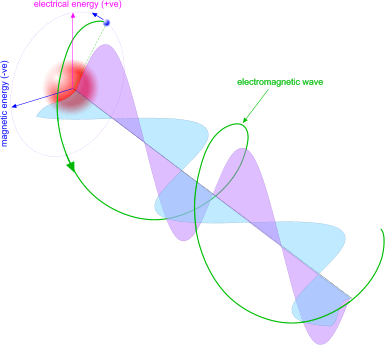
Fig 6. Electro-Magnetic Energy
ATOMS is an atomic calculator that can tell you the properties of every electron in any shell in any atom at any temperature. CalQlata believes that this is the only calculator that can do this. This calculator will be updated as more knowledge of the atom is generated.
Operation
The generation of the information in the Data Listing page of the calculator for all the electron shells can slow down the input data update process. Therefore, after each data input change, you will need to click the blue text ("List data for all electrons") above the Data Listing page if you require this listing, otherwise, the Data Listing page will only list the electron properties in shell-1
Important Note: When you select an atom for calculation, the atomic number will be updated but the RAM value will not. You must make sure that the appropriate RAM is entered for the isotope required, otherwise, some of the results will be incorrect.
Units
You may use any units you like, but you must be consistent.
This is why we recommend sticking to the basic five energy-related units for all properties:
Mass (kg or lb); Length (m or ft); Time (s); Electricity (C); Temperature (K or R)
In this way, the output data will always be as follows:
Shell: N/A
temperature: K or R
orbital radius: m or ft
electron velocity: m/s or ft/s
orbital period: s
electron spin: ᶜ/s
potential acceleration: m/s² or ft/s²
potential force: kgf.m/s² (N) or lbf.ft/s² #
kinetic energy: kgf.m²/s² (J) or lbf.ft²/s² #
potential energy: kgf.m²/s² (J) or lbf.ft²/s² #
total energy: kgf.m²/s² (J) or lbf.ft²/s² #
constant of motion: m²/s or ft²/s
energy ratio: N/A
electro-magnetic frequency: /s
electro-magnetic wavelength: m or ft
electro-magnetic amplitude: m or ft
electro-magnetic charge: C
electron microstate(t): N/A
voltage: kgf.m² / s².C (J/C) or lbf.ft² / s².C #
current: C/s
resistance: kgf.m² / s.C² (J.s / C²) or lbf.ft² / s.C² #
constant of proportionality: N/A
mass: kg or lb
density: kg/m³ or lb/ft³
specific heat capacity: kgf.m² / s².kg.K (J / kg.K) or lbf.ft² / s².lb.R #
# Please note when using Imperial units: lbf.ft/s² is generally referred to as the 'ft-lbf'. If you multiply these results by 'g' you will have the result in 'ft-pdls'.
Whilst ATOMS' default values are provided in metric units; the calculator's Help Menu offers conversions for the above constants in the associated Imperial units.
Input Data
Applies to the atom:
Ṯ₁: the temperature of the electron(s) in shell-1 of the atom
RAM: the relative atomic mass of the atomic isotope
mₑ: the mass of an electron
mₚ: the mass of a proton
G: Isaac Newton's gravitational constant
k: Charles-Augustin de Coulomb's electrical force constant
Xᵛ: electron energy velocity constant
Xᴿ: electron energy orbital constant
Y: proton charge constant
kᴮ: Boltzmann's constant
e: elementary charge unit
c: the velocity of electro-magnetic radiation
Output Data
Applies to Shell-1 only) descriptions also apply to all other shells listed below Output Data:
Ṯ: the temperature of the electron(s) in shell-1 of the atom
R: orbital radius of the electron shell
v: electron curvilinear velocity
t: electron orbital period
ω: electron spin rate in radians per second
a: potential acceleration attracting the orbiting electron to its proton
F: potential force attracting the orbiting electron to its proton
KE: kinetic energy of the electron
PE: potential energy between the electron and its proton
E: total energy in the proton-electron pair
h: Isaac Newton's constant of motion for the electron
PE/KE: energy ratio, which must always be -2 (provided for verification purposes)
ƒ: frequency of the electro-magnetic energy radiated by the proton-electron pair
λ: wave-length of the electro-magnetic energy radiated by the proton-electron pair
A: amplitude of the electro-magnetic energy radiated by the proton-electron pair
Q: charge of the electro-magnetic energy radiated by the proton-electron pair
Nₜ: microstate (temperature) of the electron
V: electrical voltage of the electron in units of energy ([potential] energy required to remove 1/e electrons from this orbital radius)
I: electrical current of the electron in units of energy (flow rate of 1/e electrons per second through adjacent atoms at this orbital radius)
Ω: electrical resistance of the electron in units of energy ([potential] energy resisting electron removal 1/e² from this orbital radius) {increases with rising temperature}
K: constant of proportionality for all atomic orbital shells, which must always be 0.15587874533403 (provided for verification purposes)
m: the mass of the atom
ρ: the density of the atom at temperature (Ṯ) {increases with rising temperature}
SHC: specific heat capacity of the atom
The listing below the output data (if selected) provides the above output data for all electron shells
Applicability
ATOMS is applicable to all atoms, at any temperature
Accuracy
ATOMS is as accurate as the input data provided
Further Reading
You will find further reading on this subject in reference publications(67)

