Polymer Database (plastics to elastomers)
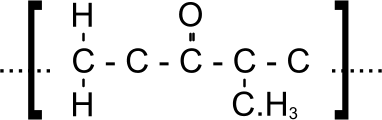
Fig 1. Typical (fictitious) Polymer Link
Polymers are naturally occurring chains of carbon atoms that have picked up other atoms, such as hydrogen, nitrogen, oxygen, etc. during - and since - their existence as cellulose molecules (plants). Each polymer comprises a chain of different, specific repeating links, which defines its properties. Fig 1 shows a typical - but fictitious - link, the fictitious polymer chain would comprise a sequence of these links. It is because we know the atomic structure of each polymeric chain that we can accurately calculate the specific heat capacity of virgin (pure) material. Moreover, we also know that the purer the polymetric material (the fewer its impurities), the lower its engineering properties; except elongation, which increases.
Almost all polymers comprise amorphous carbon chains of a particular series of links, the structure of which defines the ability of such chains to slide relative to its neighbouring chains. It is this structure and resultant slippage that defines a polymer's engineering properties.
Manufacturers extract and manipulate these links to create polymers with particular properties, sometimes deliberately, but mostly by chance. The polymers database does not include the properties associated with their manufacture, because, just as with all CalQlata's products, Polymers is aimed at those of us that use them, rather than those of us that make them.
Polymer Types
There are a number of polymer types, each of which is briefly described below:
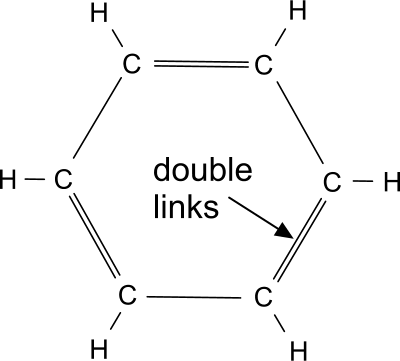
Fig 2. Typical (fictitious) Polymer Ring
Thermoplastic: a polymer chain that permits free slippage relative to neighbouring chains. As heat energy is applied to the material, the electrical repulsion between adjacent chains rises, decreasing effective magnetic attraction and thereby reducing the polymer's viscosity; it begins to melt. These polymers constitute the majority of polymers, and are also more likely to recycle.
Thermosetting plastic: imagine a thermoplastic material as described above, and then [ionically] link adjacent chains, such that relative slippage isn't possible. There you have a thermosetting polymer. As heat energy is applied, viscosity does not change. Instead, the material will char, and eventually burn. A thermosetting plastic is rigid and brittle, but can be made very strong with suitable [reinforcing] filler material. This type of material applies both to phenolics and elastomers. They can only be recycled as filler powder or chips.
Elastomer: imagine a thermosetting plastic but one with carbon-rings (Fig 2) attached. Remove an hydrogen atom or one of the double links (Fig 2) from the benzene ring and attach it to a carbon chain. The chain's irregular shape will make it much less likely to slip, even if the number of adjacent inter-chain links are reduced, making this material much less rigid than thermosetting plastics, whilst improving recoverability. Such is an elastomer (or rubber)
Thermoplastic elastomer: imagine an elastomer as described above but without the inter-chain links. As heat energy is applied, the material will eventually melt, just like a thermoplastic, but it will also have the flexible and recoverable properties of the elastomer. These elastomers may be recycled, and they also exhibit improved engineering and physical properties.
Polymers Database – Technical Help
It is important to remember that any and all polymer properties, irrespective of their source, are approximate and must be treated with caution.
Even though some polymers exhibit undeniable properties, such as the low-frictional and high-temperature characteristics of PTFE, their actual values are rarely predictable to within an accuracy of ±10%.
The only exceptions to this are the values stated in this database for specific heat capacity, which is calculated (in Polymers) for the polymer and its filler (reinforcement) using exact values for the atoms of each individual chain-link. However, these values apply only to pure, virgin material.
Units
Values are provided in both metric and Imperial units. You simply click the 'change to ...' button to select the units you require.
Data
Nomenclature
Chemical name: the chemical name provided by the BPF ⁽¹⁾
polymer type: the type of polymer as described above
abbreviation: the abbreviation recognised by the BPF for the polymer ⁽¹⁾
popular name: the 'man-on-the-street' knows these polymers by common or popular names that - in many cases - are trade-names asigned by a particular manufacturer; such as Kevlar® by Dupont ⁽¹⁾
group: the name of the group to which the polymer belongs as defined by the BPF ⁽¹⁾
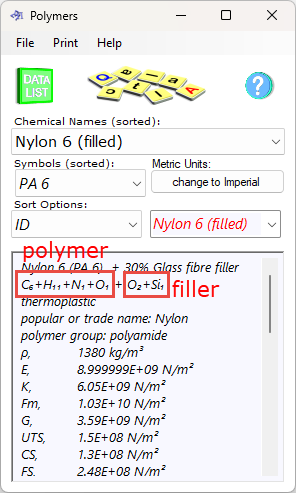
Fig 3. Chemical Formulas; polymer+filler
fill: the percentage by weight of the filler (or reinforcement) embedded in the polymer
filler: the filler (or reinforcement) embedded in the polymer to enhance its properties
polymer formula: the chemical formula for the pure polymer (Fig 3)
filler formula: the chemical formula for the pure filler material defined in Polymers (Fig 3) ⁽²⁾
Engineering Properties
density: the density of the material excluding voids. The value stated in Polymers is for the solid polymer filament, not a fabric comprising the filaments
Young's modulus: the modulus of the polymer under tensile load
bulk modulus: the bulk modulus of the polymer
flexural modulus: the modulus of the polymer under bending
shear modulus: the shear modulus of the polymer
ultimate tensile strength: the tensile stress, which if exceeded will cause disruption (tearing) of the polymer
compressive strength: the compressive stress, which if exceeded will cause permanent (plastic) deformation of the polymer
flexural strength: the bending stress, which if exceeded will cause permanent (plastic) deformation of the polymer
Izod impact (notched): the load per unit length of sample which if exceeded will damage (break) the polymer. A value of '0' in the polymer database means that the polymer doesn't break under test conditions
elongation: the expected percentage increase in length at its UTS
hardness: the Shore hardness of the polymer. Elastomers are quoted in grade 'A' ⁽³⁾, and all other polymers are quoted in grade 'D'
Poisson's ratio: the relative strain normal to that experienced aligned with the applied load
friction coefficient: the expected frictional resistance of the polymer when rubbed - under light load - against dry smooth steel ⁽⁴⁾
Physical Properties
electrical resistivity: All polymers are electrically resistant unless filled with an electrically conductive filler ⁽⁴⁾
specific heat capacity: The actual (calculated) value for the specific heat capacity of a pure polymer and its filler material
thermal conductivity: the rate at which heat passes through the polymer ⁽⁴⁾
thermal expansivity the rate at which the polymer will increase in size per degree of heat applied ⁽⁴⁾
minimum service temperature: the temperature below which, the polymer will begin to lose its engineering properties ⁽⁴⁾
maximum service temperature: the temperature above which, the polymer will begin to lose its engineering properties ⁽⁴⁾
water absorption: the expected percentage weight of water the polymer will absorb under normal conditions over a period greater than 24-hours ⁽⁴⁾
Applicability
Polymers applies only to those included in the database.
Accuracy
It is almost impossible to replicate the properties of polymers, even those of the same structure manufactured by the same people on the same equipment. This is because it is almost impossible to create a perfect (pure) polymer.
It is for this reason that the properties quoted by any manufacturer for any polymer will be approximate; they always quote a range of values. In many cases, manufacturers actually omit most of the properties in their technical data sheets because they know how unreliable these values are.
Therefore, whilst we include documented values for all of our properties, they will tend to be at lower-than-average, but should be treated with caution.
CalQlata recommends that when designing any product or appliance using a polymer, extensive testing must be carried out on the selected manufacturer's polymer and suitable safety factors duly applied.
Notes
- These may vary between sources around the world.
- Many different fillers or reinforcements may be applied to a polymer. Those selected in Polymers are predominantly glass fibre for comparison purposes. The properties of others, such as minerals and natural fibres, may vary considerably dependent upon a manufacturer's preference.
- Shore Grade-A is generally regarded as equivalent to the standard IRHD, which is normally used to define the hardness of elastomers.
- This value is highly subjective and cannot therefore be relied upon. It can vary considerably with environmental conditions, in addition to the polymer's manufacturer.
Further Reading
You will find further reading on this subject in reference publications(76)

